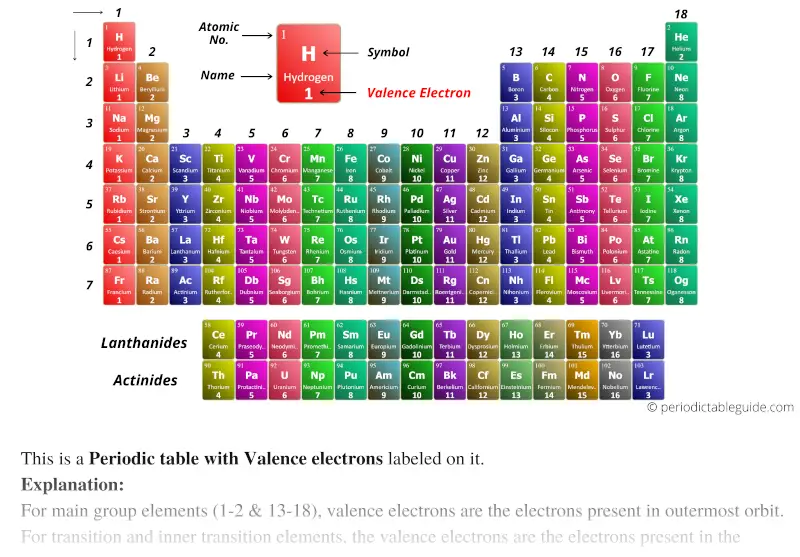
You will see in the next chapters that the chemical properties of elements are determined by the number of valence electrons. We can see from the electron configuration of a carbon atom-1 s 22 s 22 p 2-that it has 4 valence electrons (2 s 22 p 2) and 2 core electrons (1 s 2). Valence shell electrons (or, more simply, the valence electrons) are the electrons in the highest-numbered shell, or valence shell, while core electrons are the electrons in lower-numbered shells. Valence electrons Google Classroom About Transcript Valence electrons are the electrons in the outermost shell, or energy level, of an atom. Thus, it is convenient to separate electrons into two groups. The number of valence electron remains constant even when the number of shells expands as we move from the top to the bottom of a group. 
Here, we just look up the element in the periodic table to get its specific location. The path a specific element will take depends on where the electrons are in the atom and how many there are. Using the Periodic Table In order to count the valence electron of an element, this is the most used technique. Other transition metals have important functions in the body, despite being present in low amounts.\,3s^1 \nonumber \]Ī chemical reaction results from electron removal, electron addition, or electron sharing of the valence electrons of the different atoms. It is the presence of this particular transition metal in your red blood cells that allows you to use the oxygen you inhale. Because each orbital can have a maximum of 2 electrons, there are 2 columns in the s. The electron configurations of the elements are in Figure 6.9.2. Each hemoglobin molecule has four iron atoms, which act as binding sites for oxygen. Excluding the transition metals and helium, any element on the periodic table will have the same number of valence electrons as the final digit of the group. 1: The Periodic Table, Showing How the Elements Are Grouped According to the Kind of Subshell ( s, p, d, f) Being Filled with Electrons in the Valence Shell of Each Element. The crucial atom in the hemoglobin protein is iron. Hemoglobin is a relatively large molecule, with a mass of about 65,000 u. Hemoglobin combines with oxygen and carbon dioxide, transporting these gases from one location to another in the body. The critical part of the red blood cell is a protein called hemoglobin. Without red blood cells, animal respiration as we know it would not exist. Red blood cells are cells that transport oxygen from the lungs to cells of the body and then transport carbon dioxide from the cells to the lungs. The chemistry of iron makes it a key component in the proper functioning of red blood cells. Transition metals have interesting chemical properties, partially because some of their electrons are in d subshells. Because iron has relatively massive atoms, it would appear even lower on a list organized in terms of percent by atoms rather than percent by mass. The first element appearing on the list that is not a main group element is iron, at 0.006 percentage by mass. Most of the elemental composition of the human body consists of main group elements. The number of valence electrons of an element can be determined by the periodic table group (vertical column) in which the element is categorized. 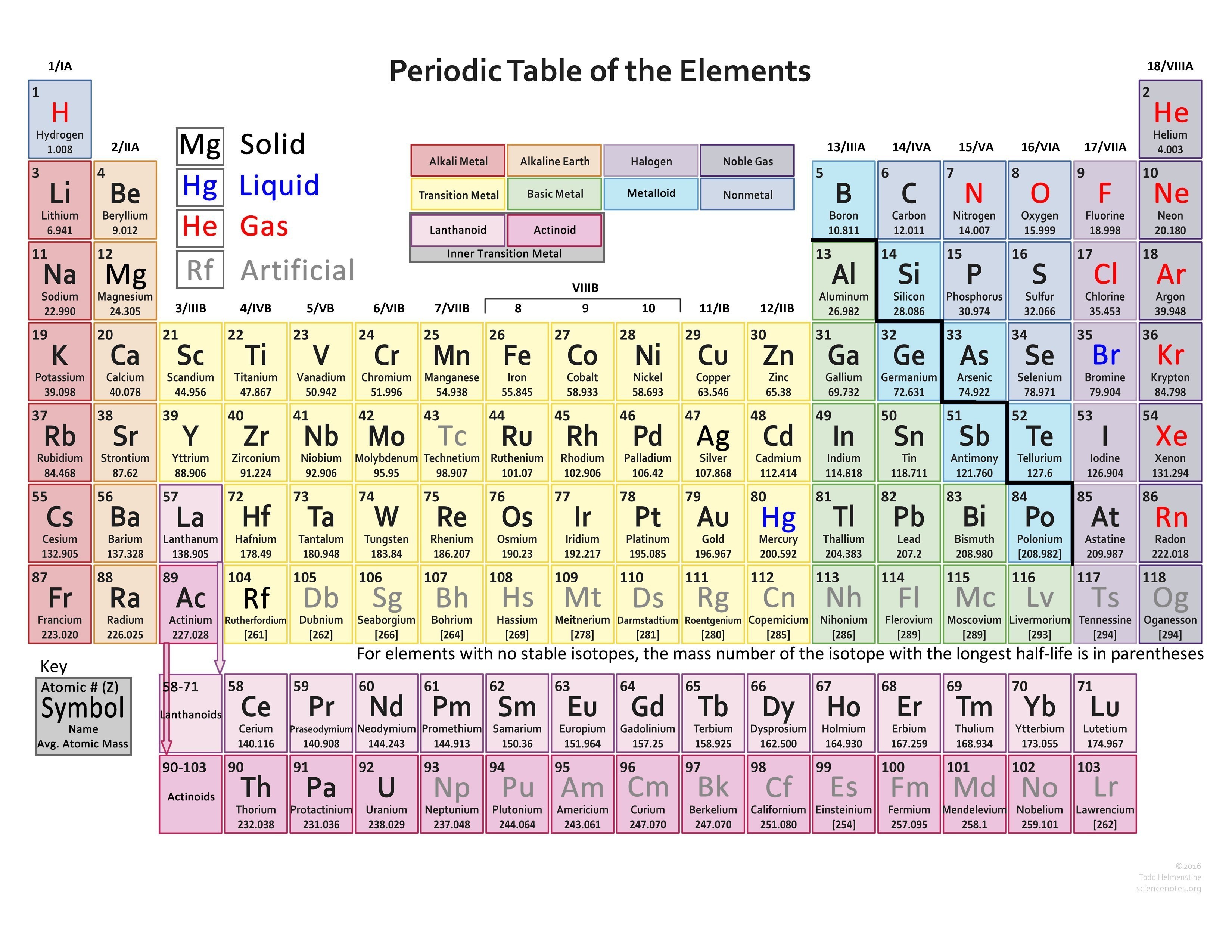
To Your Health: Transition Metals in the Body Each group is located in a different part of the periodic table. Elements are either metals, nonmetals, or semimetals.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
